 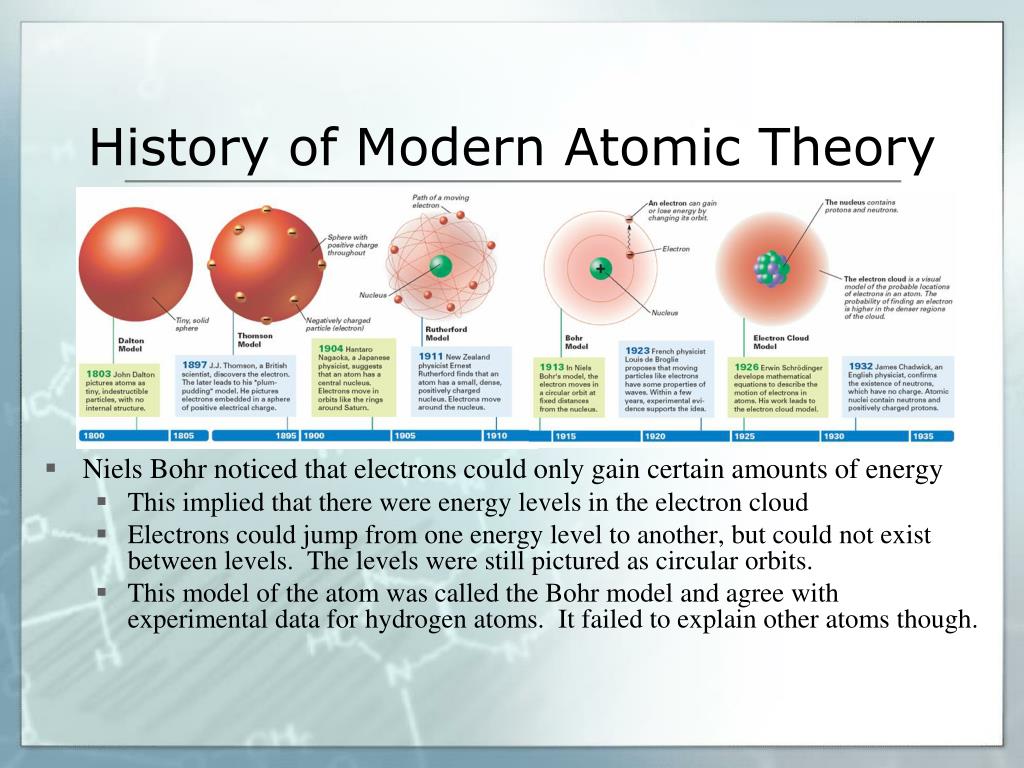
The “ electron cloud” model better represents our current understanding of the structure of the atom.The Bohr model does not depict the three-dimensional aspect of an atom, and it implies that electrons are in static orbits.Two models commonly used are the Bohr and the “ electron cloud” (Quantum Mechanics) models. 
Their understanding of the structure of the atom continues to evolve.Create and share a new lesson based on this one. Theresa Doud details the history of atomic theory. Scientists use models to help explain the structure of the atom. Atomic theory is a scientific description of the nature of atoms and matter that combines elements of physics, chemistry, and mathematics. How do we know what matter is made of The quest for the atom has been a long one, beginning 2,400 years ago with the work of a Greek philosopher and later continued by a Quaker and a few Nobel Prize-winning scientists.Many scientists have contributed to our understanding of atomic structure.Size at the atomic level is measured on the nanoscale.Atomic Theory Timeline Here is a timeline of some of the major ideas. As scientists have learned more and more about atoms, the atomic model has changed. For over two centuries, scientists have created different models of the atom. Previously, an atom was defined as the smallest part of an element that maintains the identity of that element. Atomic Theory Timeline The atomic model has changed over time. 3.1: Atomic Theory is shared under a not declared license and was authored, remixed. Each element has its own atomic number, which is equal to the number of protons in its nucleus. Atoms themselves are composed of protons, neutrons, and electrons. 4 ), is a fundamental concept that states that all elements are composed of atoms. Chemistry is based on the modern atomic theory, which states that all matter is composed of atoms. Dalton thought that atoms were the smallest units of matter tiny, hard spheres that could not be broken down any further. However, one of his underlying assumptions was later shown to be incorrect. Protons and neutrons are made up of smaller particles called quarks. The modern atomic theory, proposed about 1803 by the English chemist John Dalton (Figure 1.5.4 1.5. Daltons ideas proved foundational to modern atomic theory.The atom is the basic building block of matter and consists of subatomic particles ( proton, neutron, electron, and quark) that differ in their location, charge, and relative mass. Rutherford’s model introduced the nuclear model of an atom, in which he explained that a nucleus (positively charged) is surrounded by negatively charged electrons.the contributions of Dalton, Thomson, Rutherford, and Bohr in understanding the atom.PS.3 The student will investigate and understand the modern and historical models of atomic structure.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
